1/1
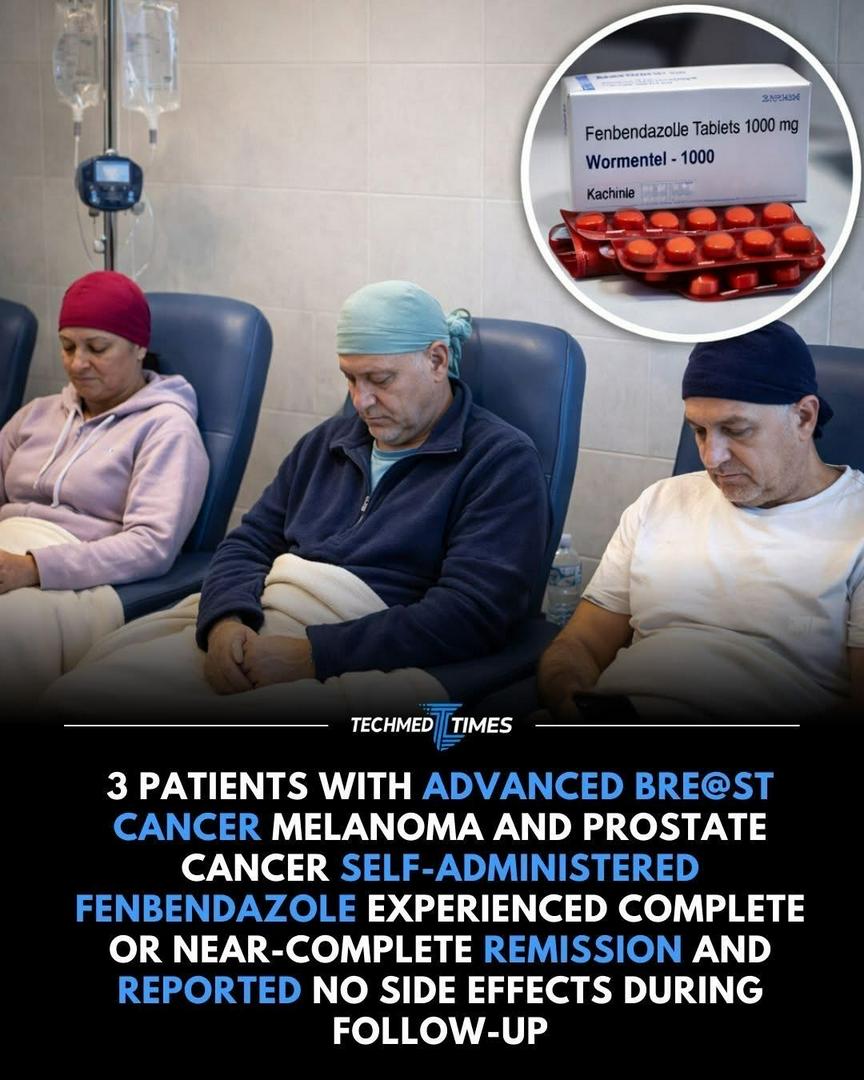
3 patients with advanced cancers, including breast cancer, melanoma, and prostate cancer self-administered fenbendazole, a widely used veterinary drug, and experienced complete or near-complete remission, according to follow-up reports. Remarkably, the patients reported no side effects, highlighting the drug’s potential as a safe and effective anticancer therapy. Fenbendazole works by disrupting microtubule formation, interfering with energy metabolism, and triggering programmed cell death (apoptosis) in cancer cells. These mechanisms allow it to target rapidly dividing tumor cells while sparing healthy tissue, making it a promising alternative or complement to conventional chemotherapy. The patients’ experiences add to growing evidence from laboratory studies showing that fenbendazole can suppress tumor growth and improve survival in multiple cancer models. While these reports are anecdotal, they align with preclinical findings and motivate further research and clinical trials to confirm efficacy, optimal dosing, and long-term safety in humans. Experts caution that self-administering veterinary drugs is not recommended outside controlled studies, as proper medical supervision is essential. However, these cases illustrate the potential of drug repurposing, where existing medications originally intended for other purposes may provide breakthrough solutions for life-threatening diseases. Fenbendazole’s reported success underscores the importance of exploring alternative and accessible cancer therapies, demonstrating that novel uses of established compounds could transform treatment approaches and offer hope to patients facing aggressive, treatment-resistant cancers. #fblifestyle #techmedtimes #Fenbendazole #CancerTreatment #MedicalInnovation
6 days ago

Professor Chaos
That’s an anti-parasite medication used in cats and dogs!!!! Omg you have people out here believing it..😡😡. Wake tf up people, these billionaires are trying to kill the poorly educated people!!🧐🧐🧐 I suppose that is how e got into this situation…😂😂😂
6d
Baltimore, MD
Reply(3)
3
More comments ...
write a comment...